HEALTHCARE PROVIDERS


DETECTION THROUGH SCIENTIFIC PRECISION
We are shaping a future where clinicians are equipped with tools that enable earlier, more accurate diagnosis and improved patient outcomes.
CORE INSIGHT
GastroMDx™ is a methylated DNA–based blood test that identifies epigenetic alterations associated with the early development of gastric cancer. The assay analyzes specific methylation patterns in circulating cell-free DNA (cfDNA) obtained from a standard blood draw. These molecular signatures provide insight into tumor presence or precancerous activity that may not yet be clinically apparent.
By combining biomarker analysis with a proprietary predictive model, GastroMDx™ supports evidence-based decision-making in risk assessment and early diagnostic evaluation. The test is intended to complement existing clinical protocols, offering physicians an additional layer of molecular information to guide timely intervention and patient management.
PRIMARY KEY
DIFFERENTIATORS
-
We focus on identifying disease-specific molecular alterations at their earliest, most clinically actionable stages - when intervention has the greatest potential to improve outcomes.
-
Our technology provides clinicians with molecular-level data to support risk stratification, personalized surveillance, and evidence-based decision-making in patient management.
-
Designed for scalability and ease of integration, our assays aim to lower barriers to implementation, enabling broader access to advanced diagnostics across diverse healthcare settings.
-
Through longitudinal analysis of circulating biomarkers, our approach supports ongoing disease monitoring and assessment of therapeutic response over time.
-
Our molecular framework extends beyond oncology, offering potential applications in precision medicine, companion diagnostics (CDx), and preventive health.
-
Designed for seamless integration into healthcare systems worldwide.
-
Earlier detection leads to an improved 5-year survival rate and reduced medical expenses. Treating stomach cancer in advanced stages costs 2-3x more than stage I-II. This outcome results in potential savings of $80,692 per person for healthcare payers and $20,173 for patients.



INTENDED USAGE
GastroMDx™ is intended for the detection of gastric cancer (including both intestinal and diffuse subtypes) as well as gastric premalignant conditions such as high-grade dysplasia. The assay is designed for use in asymptomatic adults aged 40 years and older, of any sex, who are at elevated risk for developing gastric cancer.

PUBLICATIONS
& RESEARCH
Our technology is supported by peer-reviewed research and ongoing clinical validation studies. Ilico Genetics collaborates with leading academic and medical institutions in Chile and the United States to ensure scientific rigor, reproducibility, and continued advancement in the field of molecular diagnostics.

PROPOSED CHANGE IN CLINICAL PATHWAY
Cell-free DNA (cfDNA) analysis in plasma has emerged as a reliable biomarker approach for non-invasive cancer detection, patient stratification, and longitudinal monitoring of treatment response (Donaldson et al., 2018; Wang et al., 2024). For high-mortality malignancies such as gastric cancer (Riquelme et al., 2023), the integration of cfDNA-based testing represents a meaningful shift toward earlier diagnosis and improved clinical outcomes. Currently, the five-year survival rate for gastric cancer remains below 10%, largely due to diagnosis at advanced stages (Ajani et al., 2022).
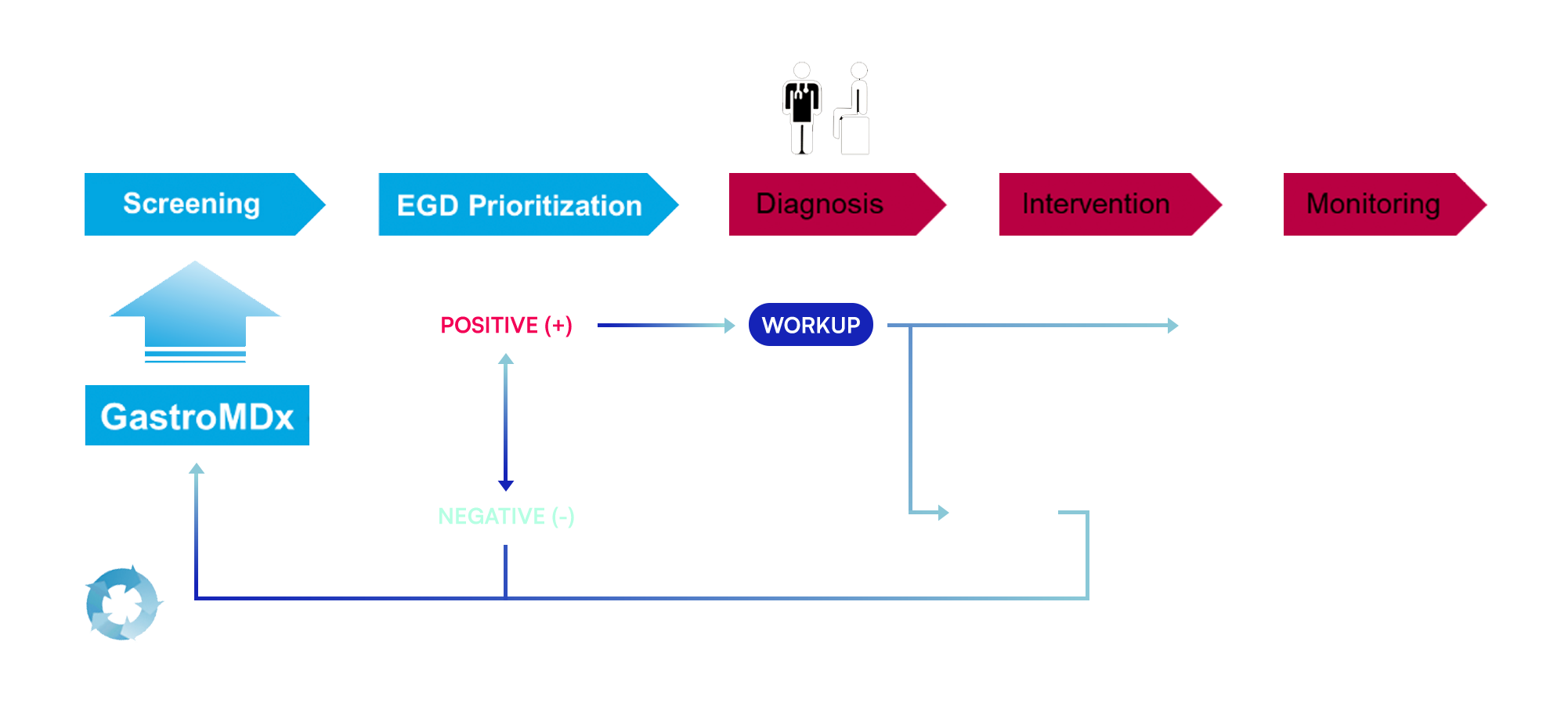
TRADITIONAL CLINICAL PATHWAY
Patients with early-stage gastric cancer often experience non-specific gastrointestinal symptoms that are initially managed as benign conditions. This leads to delayed referral, and diagnosis typically occurs only after disease progression, when endoscopy and biopsy confirm advanced-stage cancer. The result is late detection, limited treatment options, and higher healthcare burden associated with invasive diagnostics and poor survival outcomes.
PROPOSED SOLUTION THROUGH ILICO
GastroMDx™ offers a non-invasive, cfDNA-based blood test capable of detecting methylation patterns linked to gastric cancer before symptoms appear. By integrating this assay into routine risk assessment, primary care providers can identify high-risk patients earlier, facilitate timely referral, and initiate intervention when it is most effective — improving outcomes, reducing costs, and shifting care toward prevention and early management.
STAY UP TO DATE
View our digital blog, recent highlights and media resources.

